“Most hobbyists have read or heard at some point how important pH is in our aquarium. However, many of us don’t quite understand this concept or its fundamental role in the health of aquatic organisms”.
For this reason, today we are going to address this aspect so familiar yet so misunderstood of water quality, but above all, the true role it plays in our aquarium. Ready?
What is pH?

pH is the measurement of the acidity or alkalinity of a substance or solution. We use a scale that ranges from 0 to 14 as a unit of measurement. The higher a value on the scale, the more alkaline it is, and the lower, the more acidic. To interpret a measurement, we establish that a pH of “7” indicates a neutral value, while values above are alkaline and those below are acidic.
Where do we obtain those values to determine the pH?
Here’s where the explanation gets a bit complicated. When we talk about pH, we’re actually referring to the concentration of positively charged hydrogen ions in a solution, called hydronium ions.
The mathematical formula to calculate pH is the negative logarithm base 10 of the hydrogen ion activity. pH = –log10 [aH+]. In other words, a substance will be more acidic the higher its activity of positively charged hydrogen. Conversely, when there is less activity, the substance will be more alkaline.

Why is it important to know that the pH value is a logarithmic expression?
We are very familiar with measuring water temperature using the Celsius scale, where, for example, a value of 10°C is twice as cold as 20°C, as these values form a linear or geometric scale. However, if we look at the pH formula, we’ll see that we are dealing with a logarithmic expression.
This implies that a pH value of 7.0 is neutral, but at the same time, it is ten times more acidic than a pH of 8.0, one hundred times more acidic than a pH of 9.0, and one thousand times more acidic than a pH of 10.0, and so on.

This is why a slight and sudden variation in this value in the aquarium water can imply severe consequences for an organism sensitive to pH. It is very common for this mathematical property to be overlooked, causing confusion when starting to manage pH in our aquarium.
As a consequence, it is very common to hear phrases like this:
“It is important to maintain the pH more or less neutral, between 6 and 8.”
And of course, in reality, that phrase is referring to a gradient with a difference of up to 100 times greater, too imprecise for the water quality requirements in any aquarium. It’s like saying:
“It is important to keep the water warm, between 0 °C and 100 °C.”
“We must pay attention to the pH value of our aquarium, measuring it with all the precision and accuracy that is available to us”.
pH is a physical parameter.
Chemical parameters such as hardness (GH), nitrite (NO2), phosphate (PO₄³⁻), or dissolved oxygen (O2) indicate the concentration of various elements or compounds dissolved in the water, which are very helpful in determining water quality.
In contrast, physical parameters such as pH, temperature, density, conductivity, or REDOX potential indicate the state of the water; whether it is acidic or alkaline, oxidizing or reducing, hot or cold, dense or fluid, conductive or insulating, etc.
Physical parameters are also characterized by influencing all other parameters, whether physical or chemical, and pH is no exception. Let’s look at one of the most prominent examples to understand the importance of pH in our aquarium:
Influence on the toxicity of ammonia/ammonium in water.
Ammonium present in the water of an aquarium arises as a result of fish excretions and other organic waste, such as decomposing plant tissue. The presence of ammonium initiates the bacterial process known as the “Nitrogen Cycle.”
When nitrogen (N) is present in water (H2O), it can combine with hydrogen (H+ / OH–) as either ammonia (NH3) or ammonium (NH4+), depending on the pH of the aquarium. If the pH value is acidic, nitrogen combines to form NH4+, whereas if it is alkaline, it forms NH3. This is critical in determining water toxicity because ammonia is between 200 and 300 times more toxic than ammonium. (Hinton DE, Laurén DJ, 1992)

When nitrogen (N) is present in water (H2O), it can combine with hydrogen (H+ / OH–) as either ammonia (NH3) or ammonium (NH4+), depending on the pH of the aquarium. If the pH value is acidic, nitrogen combines to form NH4+, whereas if it is alkaline, it forms NH3. This is critical in determining water toxicity because ammonia is between 200 and 300 times more toxic than ammonium. (Hinton DE, Laurén DJ, 1992)
“During cycling, it is preferable for aquariums to maintain a pH below 7.0, as the ammonia levels will be elevated during the first few days”.
The exception is for aquariums whose inhabitants require alkaline pH values, such as those for saltwater, brackish water, or African cichlids. Let’s take a look:
The importance of pH on the organisms of aquatic life.
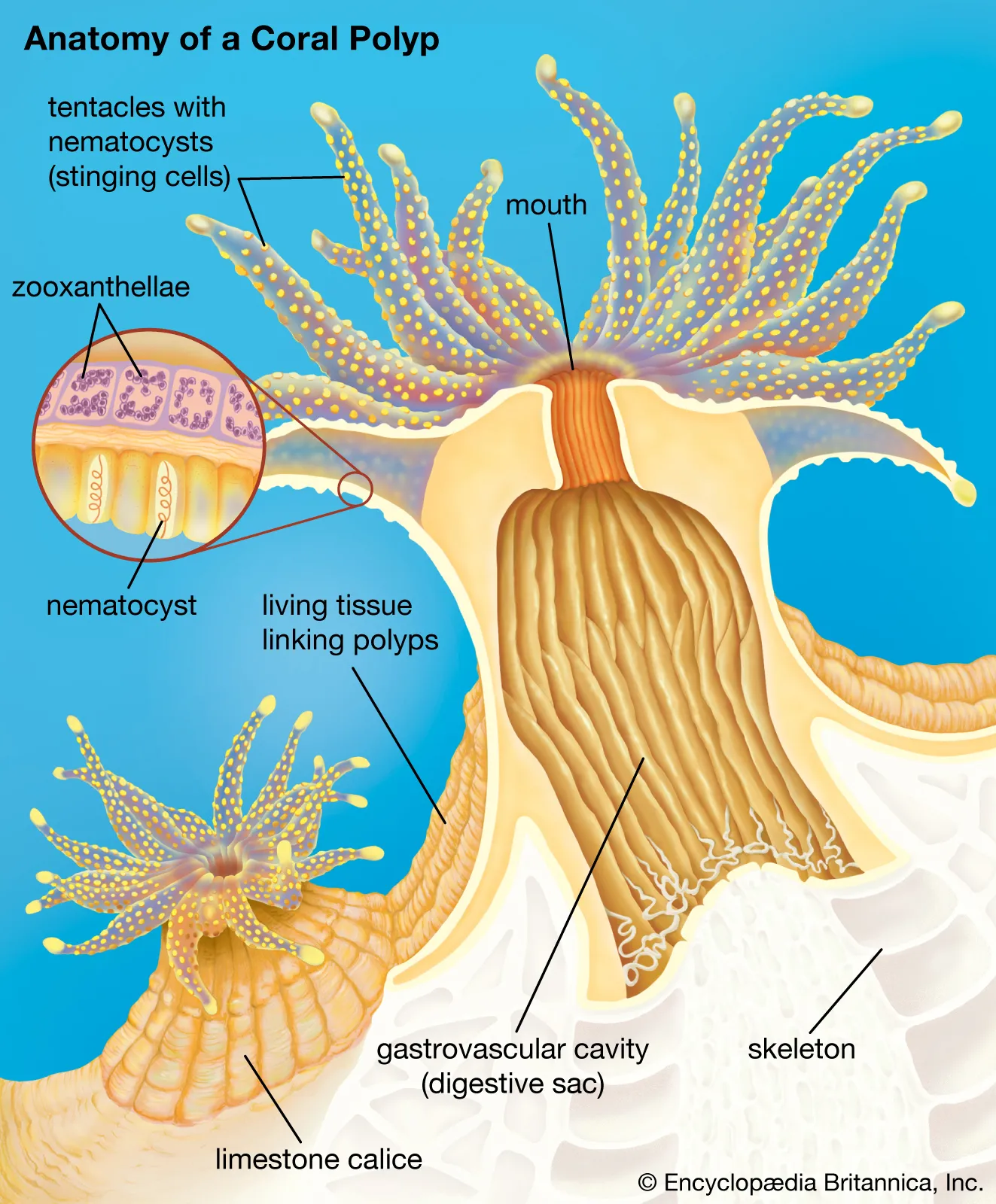
Spicules, thalli, skeletons, shells, spines, and teeth.
With rare exceptions such as tunicates (ascidians), most aquatic organisms need to produce calcium carbonate (CaCO3) to build their rigid tissues, essential for their defense and various vital functions.
However, this mineral reacts by dissolving in water when the pH drops below 7.8. This hinders the creation of new tissue and seriously weakens the already formed tissue. This decalcification syndrome is especially critical in organisms adapted to living in alkaline waters.
Fish metabolism and health.
We know that not all fish have the same tolerance to pH alterations, although we must keep in mind that if we deviate from this range, health problems will soon appear. Let’s see why:
In the medium term, a pH that is too acidic leads to problems with calcium deposits in their kidneys (nephrocalcinosis) and decalcification in hard tissues. It also affects growth due to problems with appetite and nutrient absorption.
“The most immediate effects of an inappropriate pH value on fish are the reduction of their respiratory efficiency and the capacity of their osmoregulatory system”.
The acidosis syndrome.
In the short term, a pH that is too low causes a drastic decrease in the blood hemoglobin’s ability to absorb oxygen (Bohr effect, Wedemeyer, 1996). This leads to an excess of CO2 in the blood (hypercapnia) with a consequent decrease in pH (fish blood has a range between 7.7 and 8.0).
As a result, the affected fish may become lethargic, gasp at the water surface, and exhibit accelerated breathing.

Another relevant consequence is the increase in the inefficiency of their osmoregulatory system, leading to disorientation, imbalance, and loss of appetite. It is common to observe irritation in the gills and scales, inflammation and clouding of the cornea, as well as an excess production and shedding of mucus on the skin.
All this metabolic disorder, accompanied by inevitable anoxia, causes severe stress to the animal, further complicating its symptomatic picture with the fall of its immune system. (Wedemeyer G. 1970) In extreme cases or low tolerance, sudden death of the fish may occur.
On the other hand, when the pH increases too much, we face the syndrome of alkalosis. Several of the effects on metabolism are similar to those of acidosis, including their associated symptoms: gasping at the water surface, discoloration, skin irritation, cloudy eyes, and excess mucus. These symptoms highlight the importance of pH in our aquarium.
Furthermore, a pH that is too high may be accompanied by reddish gills, swollen body, frayed fins due to irritation, and erratic swimming with occasional spasms, resulting from a drop in blood potassium (hypokalemia). (Colin J Brauner, 1990) In severe cases, we may observe rapid movements or even jumping on the surface, caused by high stress.

The pH in a planted aquarium.
During the day, aquatic plants absorb carbon dioxide (CO2) and release oxygen (O2) during photosynthesis, raising the pH. Conversely, at night, they reverse this process, respiring O2 and releasing CO2, which reacts with water to form carbonic acid (H₂CO₃), lowering the pH.
In a confined environment like an aquarium, the metabolism of aquatic plants will severely modify the aquarium’s pH in 12-hour cycles if there aren’t enough buffering minerals like carbonates (KH). The importance of pH in our planted aquarium is even more critical if we intend to succeed in our project.
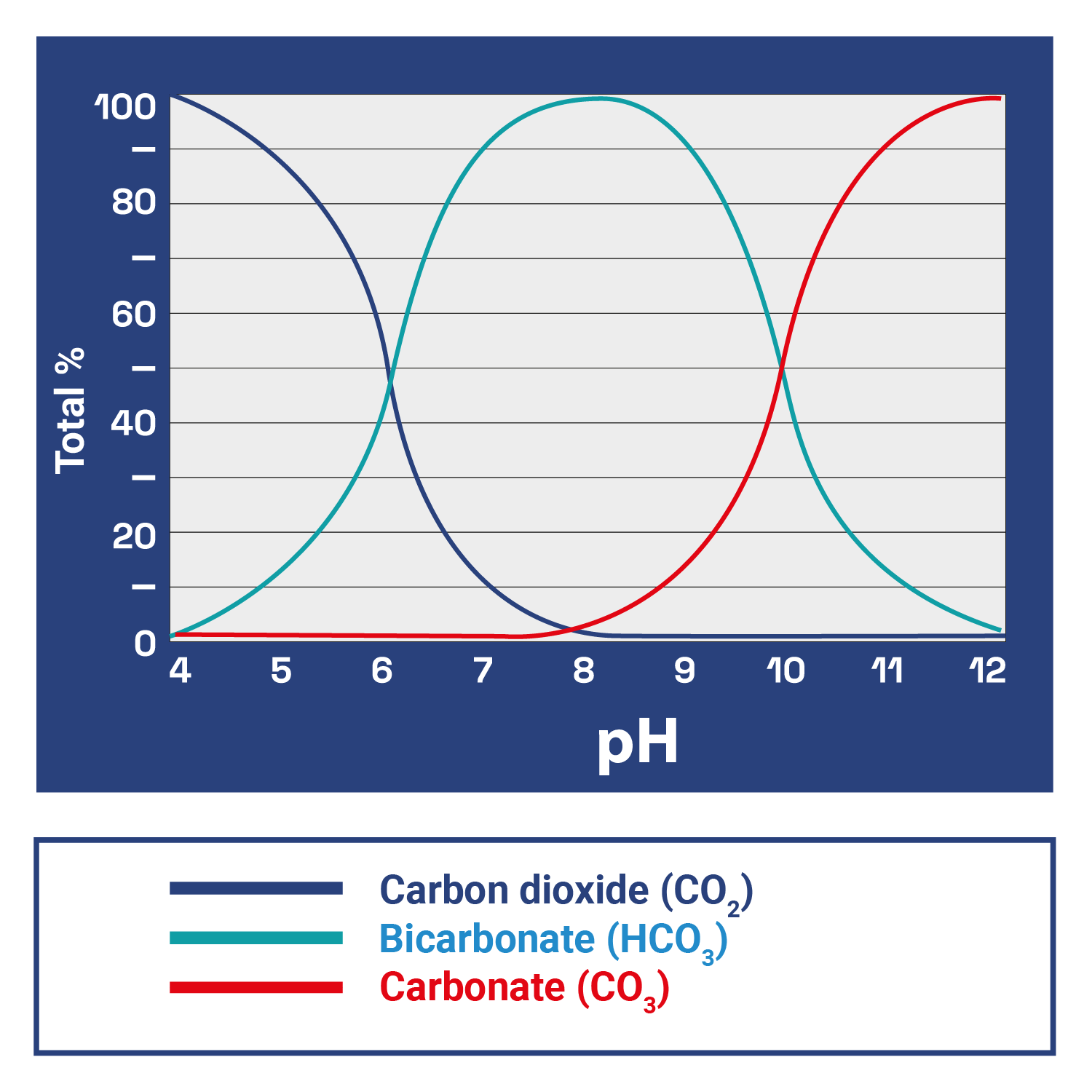
The carbon needed for photosynthesis is consumed from various sources, such as fish respiration, organic decomposition processes, and dissolved carbonate salts in the water (KH), all under a delicate and complex balance, inexorably governed and conditioned by pH:
At low pH, bicarbonate (HCO3) and carbonate (CO3) convert to CO2. At neutral pH, most of the CO2 converts to bicarbonate (HCO3), while carbonate (CO3) predominates at high pH.
Thus, by adjusting the pH value, we can control the amount of CO2 available, which is essential for the development of aquatic plants.
“Sea water has a stable pH that hardly varies from 8.2. This means that organisms living in these environments have not developed much resistance to variations in this value”.
The importance of pH in our marine aquarium.
The chemistry of seawater is usually much more complex and mineral-rich than that of freshwater rivers or lakes.
When minerals are in perfect balance in a marine aquarium, the pH ranges between 8.0 and 8.3. However, due to the metabolic action of fish, corals, and other organisms, the pH of an aquarium is altered by the quantity, type, and proportion of dissolved carbonate salts that form the KH (carbonate salts, bicarbonate, and borate).
The reason is that these carbonates tend to precipitate with dissolved calcium (Ca+) to form calcium carbonate (CaCO₃), but they can also precipitate with magnesium (Mg) to form magnesium carbonate (MgCO₃). These reactions occur when there is an excess or deficiency of any of these salts, causing a chain of precipitations that alter the pH of the water.
“Nearly all marine organisms need to develop in a pH higher than 7.8 to build their skeleton, spines, spicules, shells, and other structures”.
Below this value, CaCO₃ reacts with water, dissolving, leading to severe consequences for almost all marine organisms. In reef aquariums, we often maintain many organisms with a particularly high demand for calcium to build their skeletons or shells, such as corals of the genus Acropora spp. or mollusks of the genus Tridacna spp.
These species also require to remain in a very narrow and stable pH gradient between 8.20 – 8.30, or else they begin to lose their ability to fix calcium carbonate and develop.
Despite the fact that appropriate pH in saltwater is even more critical than in freshwater, we have a great advantage: the alkaline condition of seawater allows CO2 to penetrate it when aired at the surface, helping to balance the pH if the KH is at a value of at least 7° dKH (2.5 meq/l).
One of the most common techniques to aid in the treatment of saltwater fish infected by parasites is to give them a bath in freshwater for between 5 and 10 minutes. Similarly, freshwater fish can also undergo osmotic shock therapy, but in this case using seawater.
Fish generally tolerate a very radical change in salinity for a limited time, but this is not the case with other parameters such as pH or temperature. In fact, if we do not match the pH to the value maintained in the aquarium water from which the fish to be treated come from, they could worsen or die just from that pH difference.
We provide this example to emphasize the importance of pH in our aquarium. We should not make abrupt pH changes, as fish generally tolerate a very large change in salinity for a few minutes, but not an abrupt change in pH, even by just a few tenths in many species. This is due to the logarithmic nature of the pH scale.
The appropriate pH for freshwater and saltwater fish.

In contemporary aquarium keeping, we have access to freshwater fish from all five continents. As we have seen, each species has a range between the maximum and minimum pH for healthy development.
That’s why we must educate ourselves about these requirements before acquiring fish and combining them in our aquarium. In aquarium keeping, it’s very common to maintain what are known as community tanks.
The organisms we keep in these tanks are selected because they share a pH range that approximates 7.0 (for example, between 6.5 and 7.5). Fortunately, many of our freshwater fish, invertebrates, and aquatic plants tolerate this range quite well.
Other more advanced hobbyists, aware of the importance of pH in our aquarium, prefer to combine organisms belonging to a specific geographical area, river system, lake, or even a particular biotope, which share a similar water quality. This has a clear advantage, as it allows them to be much more precise with an appropriate pH, enjoying healthier and more vibrant fish.

Marine fish and corals collectively benefit from sharing a pH range between 8.0 and 8.3. This allows us to focus solely on stabilizing this value.

Alright, pH is important. But, how do we measure it?
Now that we have a better understanding of the importance of pH in our aquarium and its decisive influence on aquatic organisms, it’s time to tackle the easier part: measuring it. The current market offers us various methods for measuring pH, all of which are very easy and quick to use. Let’s analyze the advantages and disadvantages they present to help you choose the most suitable one.
Colorimetric methods: This method involves obtaining a color on a liquid or paper strip to then be compared with a standard.
By finding the closest color match with the sample, we obtain our measurement. The two most common methods are measurement test strips (such as litmus paper) and liquid indicator kits. Test strips have the disadvantage of low precision and rapid expiration, which further reduces accuracy.
In contrast, liquid indicators are much more practical and faster. Just dissolve a few drops in a certain amount of water in a test tube and compare the resulting color with a colorimetric chart.
In a few seconds, we will know the result. It is important to acquire a test of good quality, offered by a trusted and recognized brand such as AZOO pH tests.
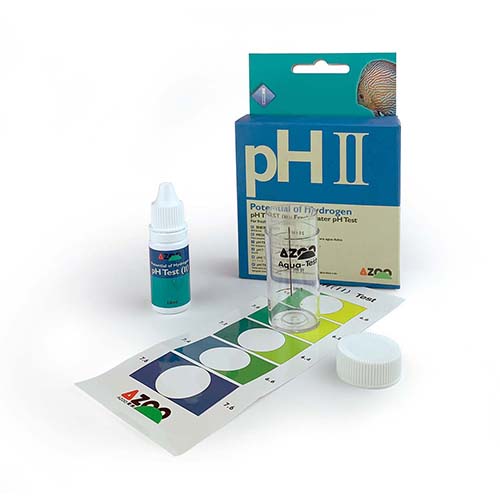
Due to the importance of pH in our aquarium, it is crucial to have a certain guarantee of accuracy and reliability when measuring it. Let’s not forget that pH is a value whose accuracy is vital. These test kits are economical and reliable. Within colorimetric tests, we will make a special mention of continuous pH monitoring tests.
Then we have the electronic measurement method. This system is by far the most accurate; we just need to calibrate it regularly and properly maintain its sensor. Apart from its great precision, another advantage is its speed and the possibility of making multiple consecutive measurements.

These devices have a glass-tipped probe filled with electrolytic solution, which we call a sensor. Its purpose is to measure the activity of hydrogen ions by generating a small amount of voltage in the sensor and reference tube of the probe.
The voltage meter converts this into a pH value and displays it on a digital screen. Although its price is higher, it more than compensates with its convenience, immediacy, and high precision. The more times we need to measure, the more cost-effective this method becomes.
There are even continuous measurement models, equipped with a controller to measure and maintain the desired value through automation.
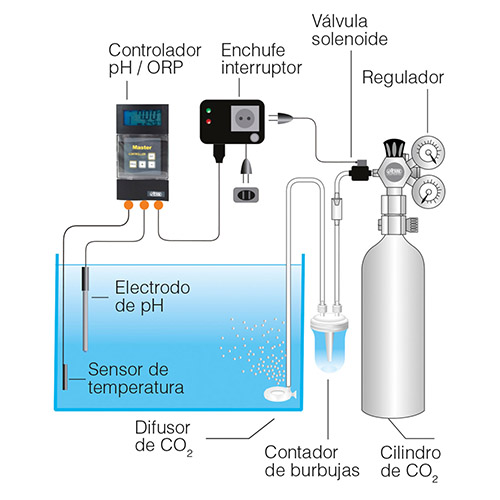
Okay, measuring pH is very easy. Now, how can I control and stabilize it in the aquarium?
If you’ve made it this far in your reading, congratulations. You’re more than ready to take control of the pH in your aquarium. To control pH, the best way is to start by measuring it. Then, we’ll add an appropriate and safe pH conditioner to the water to correct it easily. For this purpose, the market offers us two options: pH conditioners for raising or lowering pH, and buffers.
Method of buffer-type conditioners. (The concept “Buffer” is an anglo-saxon term for the chemical concept of “buffer” or “buffering”). These additives aim to give the aquarium water a “buffering” effect, so that the pH remains very stable at a specific value. To achieve this, they alter the water’s pKa factor (pKa is a measure of acidity strength).
This method involves applying a formula that combines the same salt in its acidic and alkaline forms. By combining them in the correct proportion, the manufacturer obtains the pKa factor that will stabilize the pH where we want it.
The buffer technique is extremely simple, as not only do we achieve the desired pH, but we also maintain it at that value for several weeks. Once pre-dissolved in a little water, we can add it to the water we are preparing for a partial change or directly to the aquarium. All this without running a great risk of overdosing the additive.
The most interesting aspect of these “buffers” (and one that few hobbyists know) is the ability to combine them to obtain and stabilize the pH value we want in each situation.
The other alternative method is a bit more advanced. If we want to lower the pH, we can use a conditioner based on a combination of acidic salts. This compound will react with carbonate salts (KH) resulting in carbonic acid (H₂CO₃), which will cause the pH to decrease. We just need to be careful not to overdose this additive and lower the pH too much. Many manufacturers, aware of the importance of pH in our aquarium, have developed various solutions to control this aspect.
Some advanced hobbyists use very strong acids based on phosphorus or sulfur to lower the pH, but it’s a much more aggressive and risky method of overdosing. Additionally, these acids are extremely aggressive upon contact with the skin and eyes, potentially causing severe chemical burns.
Personal protection is necessary for their use, along with prior experience and knowledge in handling them if we don’t want to cause an accident to ourselves or our aquarium. On the other hand, it’s easy to cause algae outbreaks with these compounds due to the nutrient imbalance they can cause.
If our pH is too low, it’s very likely that the cause is very low alkalinity (KH). The most reliable method is once again to avoid “home remedies.” In this case, we recommend adding a balanced solution of salts that increase carbonate hardness in a stable and controllable manner with a conditioner like AZOO Plus Aquaguard.
Conclusions:
From this article, we can highlight the following key points to understand the importance of pH in our aquarium:
- The pH indicates the acidity or alkalinity of the water, based on the concentration of positively charged hydrogen ions in a solution. To measure it, we use a scale where the minimum is “0” as acidic, “7” is considered neutral, and the maximum alkaline value is “14”.
- pH is a logarithmic and non-linear value, so when measuring it, we must be as precise and accurate as our means allow.
- pH is definitely the most important parameter of the aquarium along with the dissolved oxygen rate and temperature, as it directly affects the metabolism of aquatic organisms in the short term.
- pH directly affects the toxicity of ammonia, so it is much safer to stay in a slightly acidic pH in the presence of this nitrogen compound.
- Not all aquatic organisms have the same tolerance to pH alterations. We must find out what is the maximum and minimum gradient they are adapted to in order to avoid going beyond it.
Within their own gradient, the metabolism of organisms suffers more from sudden changes in pH than from remaining at one extreme. Therefore, we must offer sufficient time to allow for their acclimatization. As a reference, it is advisable to increase or decrease half a pH point every 24 hours. (F. Castelló Orvay, 1993) - It is preferable to measure, control, and stabilize the pH rather than wonder if the symptoms of a fish or coral are due to acidosis or alkalosis. Don’t you agree?
- The most practical and economical method for measuring pH is to use a reactive measurement kit. Electronic pH pens are highly recommended despite their higher price. They likely cost less money than the fish we keep.
- The safest and most practical way to control and stabilize the aquarium’s pH is to use high-quality commercial conditioners. We can directly modify the aquarium’s pH, avoiding causing too abrupt alterations, but the best way is to prepare the water chemistry before performing a partial change.



